The search for a COVID-19 vaccine started not long after it became clear that the disease was rapidly affecting millions of people around the world. It has been evident that there is a need to save as many lives as possible. But as infection rates continued to soar, many had still asked: “When will a vaccine for COVID-19 be available?” Now, due to the hardworking efforts of several organizations, researchers, and developers around the world, COVID-19 vaccines are available and authorities are determining how to administer as many doses to the public as possible.
Messenger RNA (mRNA) Vaccines
With the exception of mRNA vaccines, vaccines work by placing a much weaker version of the virus into our body as it helps train our immune system to produce the necessary antibodies that are needed to fight the disease. However, messenger RNA (mRNA) vaccines work by training our cells to make a protein without using the live virus. It is this protein — rather than a weaker version of the disease — that activates the immune system response in our bodies.
COVID-19 mRNA vaccines carry instructions on how our cells can produce what is known as a spike protein that is also found on the surface of the COVID-19 virus. After our cells produce the spike protein, it then breaks down the instructions and begins removing traces of the virus from our body. Once the immune system notices that the protein produced is foreign, a response is triggered to help in fighting future infection.
The use of mRNA vaccines in the fight against COVID-19 is a noteworthy accomplishment because the vaccines do not carry the SARS-CoV-2 virus. Those who get vaccinated will not get sick with COVID-19 because of the vaccine.
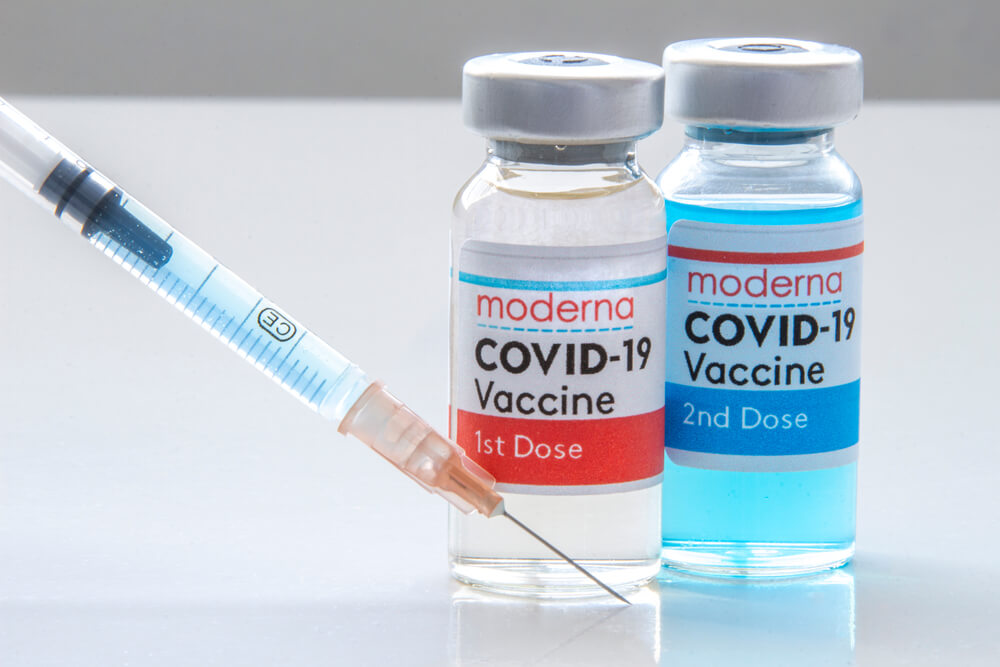
Pfizer-BioNTech and Moderna COVID-19 Vaccines
Both Pfizer-BioNTech and Moderna have developed vaccines in the fight against COVID-19 and Florida has procured doses from both companies. While both vaccines are mRNA based and are of similar effectiveness, there are some differences between the two vaccines.
The major difference between both of these vaccines lies in how they are going to be stocked. To remain effective, the Pfizer-BioNTech vaccine needs to be stocked at about -80 Fahrenheit while the Moderna vaccine only needs to be stocked at about -10 Fahrenheit. This difference is an important consideration for vaccination centers as it requires specific technology that allows for proper temperature storage.
Another difference is the age groups that can receive the vaccine. The Moderna vaccine is only available for those who are older than 18 while the Pfizer-BioNTech vaccine can be administered to those who are 16 and older.
While both vaccines require two shots before you are fully vaccinated, there are differences in the timing of each dose with each vaccine. The second dose of the Moderna vaccine is administered 28 days (one month) after receiving the first dose. The second dose of the Pfizer-BioNTec vaccine is given 21 days after receiving the first dose. It’s important to note that you cannot have one dose or the other, meaning that if your first dose comes from Moderna, then your second dose must also be a Moderna COVID-19 vaccine. The same applies with the Pfizer-BioNTech COVID-19 vaccine.
According to the Florida Division of Emergency Management, if you are able to get vaccinated in the earlier phases, the vaccine you receive will depend on the health facility that you visit as some health facilities might only have one or the other. However, as the demand for the vaccine reduces and it becomes more accessible to the public, individuals will be allowed to pick which brand of the COVID-19 vaccine they would like to receive.
Who Has Access to the Vaccine?
Since the U.S. Food and Drug Administration (FDA) issued an Emergency Use Authorization (EUA) for the Pfizer COVID-19 vaccine — granting the vaccination of individuals who are 16 years and older — Florida has taken considerable steps towards securing and distributing COVID-19 vaccines. After the announcement by the FDA, vaccines were distributed to hospitals with adequate storage capabilities, long-term care facilities, and different county healthcare departments.
According to Florida Department of Health Guidelines, the state will prioritize individuals who are 65 years and older, health care personnel with direct patient contact, the residents and staff of long-term care facilities, and people who are extremely vulnerable to COVID-19 (determined by hospital providers). Doses of the vaccine have already been shipped to all 67 county health departments and to complement this, the governor also signed an executive order to supplement current Florida vaccine laws which would allow people who are aged 65 and older, healthcare personnel, and long-term care facility staff and residents to be included in Florida’s first vaccine priority phase group.
What are the Different Vaccination Phases?
The state of Florida has drafted a three-phase COVID-19 vaccination policy. Following the COVID-19 Vaccination Program Interim Playbook for Jurisdiction Operations, this strategy follows the various phases which have been outlined by the CDC.
- Phase 1: Potentially Limited Doses
- Phase 2: A Large Number of Doses Available, Supply Likely to Meet Demand
- Phase 3: Likely Sufficient Supply, Slowing Demand
Phase 1: Potentially Limited Doses
In this phase, there is a chance that there will still be a limited supply of COVID-19 vaccine stocks. Because of this, the state would have to prioritize which groups of people will receive the vaccine as it would not be possible to vaccinate the whole population at this time. As more vaccines become available, the state will continue to follow CDC guidelines by expanding to other priority groups in the following closed point of dispensing (POD) areas:
- Hospital Closed PODs
- Long-Term Care Employees and Resident PODs
- First Responder and Critical Infrastructure Closed PODs
Phase 2: A Large Number of Doses Available, Supply Likely to Meet Demand
In this phase, there will be more mass public vaccination clinics to guarantee that the vaccine will be distributed equally, much like how COVID-19 testing centers were set up. More vaccines will become available to immediate care providers, pharmacies, and pediatricians. In the second phase, the new vaccination areas will include:
- State Managed Vaccination Sites
- Hospital Open PODs
- Public Mass Vaccination Sites
- Established Children/Adult Vaccination Centers
Phase 3: Likely Sufficient Supply, Slowing Demand
In this step, the vaccine should have become broadly available and the demand should be much less. The state of Florida will then begin to provide vaccines through frequent health care delivery systems including commercial pharmacies.
Florida Immunization and COVID-19
In preparation for the COVID-19 vaccination, the Florida Department of Health has been working on readying Florida SHOTS, a free statewide, centralized online, immunization information system (IIS). Florida SHOTS will aid all healthcare providers to keep an eye on COVID-19 immunization records in Florida. Additionally, private practices can enroll with Florida SHOTS to offer vaccination once it is available. Before any healthcare provider or facility is able to administer a COVID-19 vaccine, they must be registered with Florida SHOTS first. The major benefit of the program lies in the centralization of vaccine records in Florida.
Florida SHOTS will also be crucial in sending out reminders for the second doses of the mRNA vaccines which are being issued. The program has a readily available reminder-recall function for vaccination providers. This will ensure that people do not miss the second dose of their COVID-19 vaccine.
The TopLine MD Alliance encourages all affiliated providers to receive the COVID-19 vaccination series as soon as it is available. Practices that plan to offer vaccinations for staff or patients are proactively registering with Florida SHOTS in order to be eligible to do so when the vaccine is available. For more information on COVID-19 and available vaccines, visit www.toplinemd.com/COVID-19.
The TopLine MD Alliance is an association of independent physicians and medical practice groups who are committed to providing a higher standard of healthcare services. The members of the TopLine MD Alliance have no legal or financial relationship with one another. The TopLine MD Alliance brand has no formal corporate, financial or legal ties to any of the affiliated physicians or practice groups.