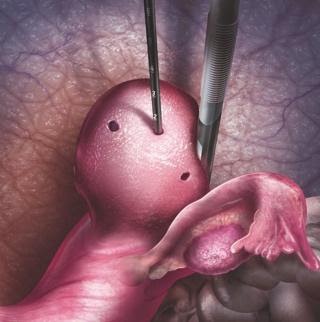
Acessa Procedure for Fibroids
Who is this procedure for?
This procedure is for pre-menopausal women (typically younger than 55 years old) who are tired of living with the ongoing pain and discomfort of fibroids and are looking for a minimally invasive option that allows them to keep their uterus and return to daily life quickly after the procedure.
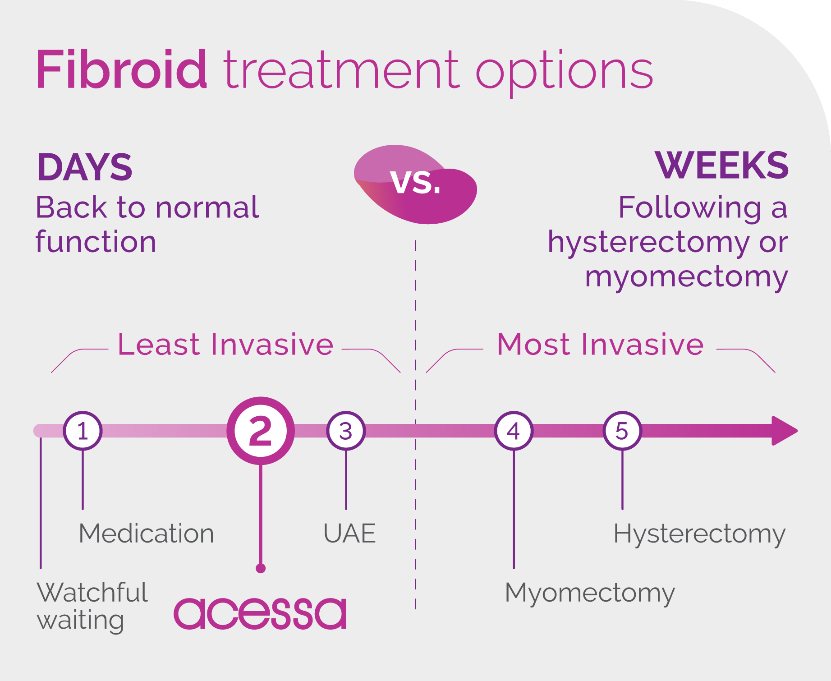
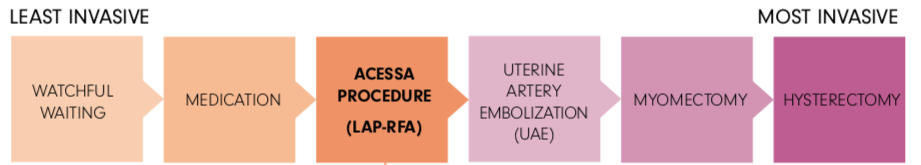
How big of fibroids can Acessa treat? What types of fibroids can Acessa treat?
Size and location of fibroids are important factors in what procedure is right for you. That’s why physicians do a MRI or ultrasound prior to determining if you are a candidate for the procedure.
The Acessa procedure has been studied on fibroids up to 10cm, commercially physicians have treated larger fibroids. The procedure is not recommended for pedunculated fibroids (fibroids on a stalk).
Telephone
How does Acessa compare to other treatment options?
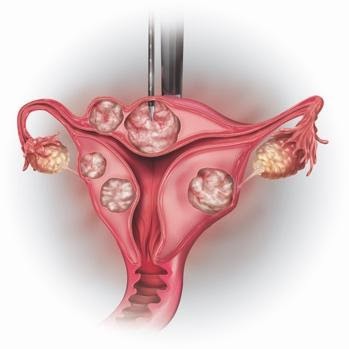
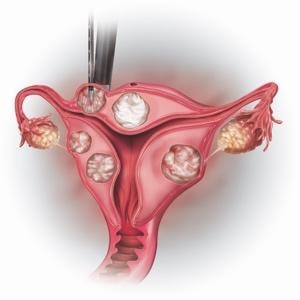
How the procedure works?
- The clinical terminology for the procedure is Laparoscopic radiofrequency ablation (Lap RFA) for fibroids.
- The Acessa procedure typically takes between 90 to 120 minutes depending on the size, location, and number of fibroids.
- Fibroids are heated, using radiofrequency ablation, to the point where they are destroyed and are no longer the consistency of a fibroid. The heat denatures the proteins inside the fibroid cells.
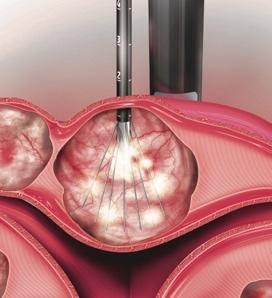
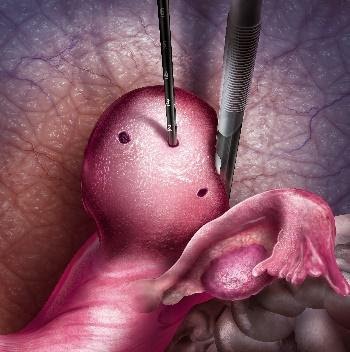
- After being heated they go from the consistency of a hard baseball to a soft marshmallow

- The change in the consistency to a soft marshmallow and gradual shrinkage is what creates relief from the fibroid symptoms.
- When the heated tissue shrinks into the marshmallow the reduced cells typically get recycled into the body. This is the same natural process the body uses when recovering from a scab.
- There is no incision within the uterus which allows the physician to spare the healthy tissue.
- This procedure can also be used in conjunction with other procedures such as a myomectomy and endometrial ablation.
FAQ
The Acessa Procedure – Results
- Addresses nearly all symptoms such as excessive bleeding, prolonged periods, urinary frequency, painful sex, stomach, lower-back and pelvic pain. Individual results vary based.
- In clinical studies, there was an average shrinkage of 45% in fibroid size post-procedure. This number is contingent on where the fibroid is located and the size.
- Women typically see the most symptom improvement within 3 months of the procedure with continued improvement throughout the first year.
- Studies have shown both clinically and statistically significant reduction in period blood loss.
- Significant reduction in fibroid and uterine volume.
BY THE NUMBERS:
- 82% of women had lighter periods.
- 94% patient satisfaction rate.
- Only 11% of patients required additional reintervention after the Acessa procedure.
Why haven’t I heard of Acessa before? Is it brand new?
Laparoscopic RFA was first performed on fibroids in 1999 by Dr. Bruce Lee. (2) After many successful studies, the original Acessa System was FDA cleared in November 2012. Since 2012, physicians have performed over 3,000 procedures to date.(5) The newest, most advanced technology, the Acessa ProVu system, was FDA cleared in 2018.
Your physician can explain the potential complications of the Acessa Procedure, as well as those of other available fibroid therapies.
Is it covered by insurance?
Acessa is covered by many insurance carriers. The first step if you are interested in Acessa is to contact a physician who offers Acessa and schedule an initial consultation. After you have a consultation with a physician and determine if you are a candidate for the procedure, they can help you understand the benefits and coverage for Acessa.
Disclaimer
Acessa Health does not make any recommendations, referrals, or endorsements regarding specific physicians with whom patients may seek treatment, nor does this site serve as a tool for verification of a specific surgeon’s credentials, qualifications, or abilities. Only a trained physician is qualified to recommend treatments and/or make diagnoses. You assume full responsibility for your communications and interactions made with any physician you choose to contact from the use of this website.
RISK STATEMENT
Acessa is cleared by the FDA for the treatment of symptomatic uterine fibroids. There are risks associated with all minimally invasive surgical procedures, including serious complications such as infection, bowel injury, and postoperative bleeding. Insufficient data exist on which to evaluate the safety and effectiveness of Acessa procedure in women who plan future pregnancy. Therefore, the Acessa procedure is not recommended for women who are planning future pregnancy. Please consult with an Acessa-trained gynecologist to understand the risks of surgery and find out if Acessa may be right for you.
Patient Feedback
Marina D.
Dr. Stine is the best Gyn I’ve ever had in all my 53 years! He’s caring and compassionate and listens to me and has the highest professionalism and ethics. His skills in the OR are superb from my personal experience providing me with a newly found quality of life! I have recommended him to my family and will also be taking my daughters to him. That’s how much trust I have in him!
Valerie S.
Dr. Stine is the absolute best! He is thorough, so comforting and knows his stuff! He truly cares about his patients and takes such great care of us. He is a blessing to all his patients.
Melissa D.
My experience with Dr. Stine was basically perfect! I know a lot of women, myself included, that are not comfortable with a male doctor. Dr. Stine changed that for me!
I felt so at ease with him, his bedside manner is amazing. His surgical skills are TOP NOTCH! Do yourself a favor and become a patient of his!
Request an Appointment
Have a question about our practice or want to schedule an appointment? Reach out and speak with a member of our team.